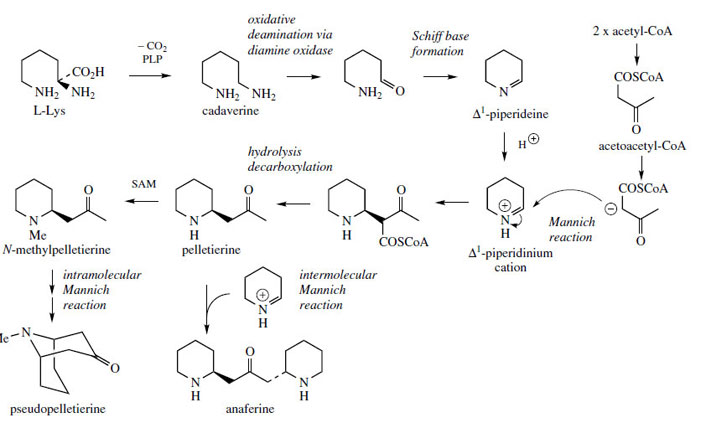
The influence of the introduction of chiral center in piperidine ring... | Download Scientific Diagram
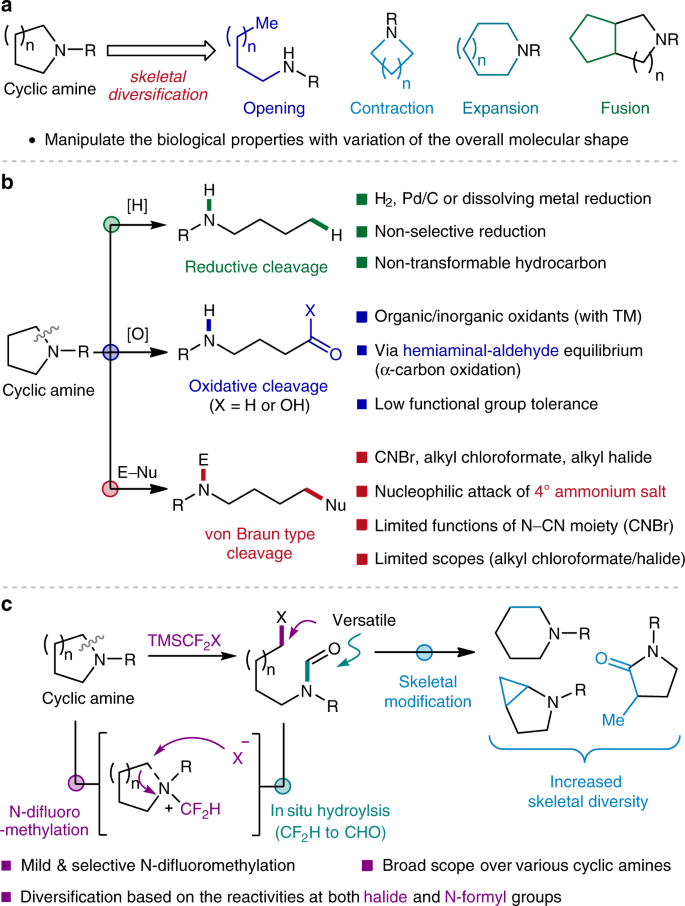
Ring-opening functionalizations of unstrained cyclic amines enabled by difluorocarbene transfer | Nature Communications

Some compounds containing a piperidine ring in their structures. (A)... | Download Scientific Diagram
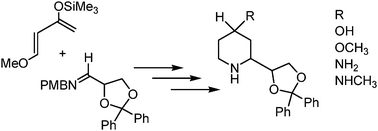
Synthesis and NMDA receptor affinity of dexoxadrol analogues with modifications in position 4 of the piperidine ring - MedChemComm (RSC Publishing)
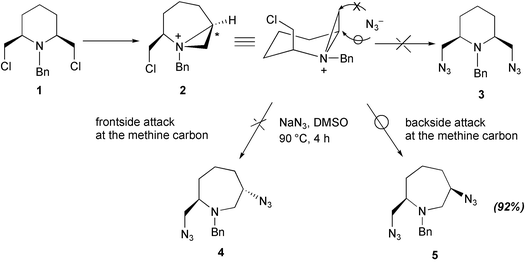
Stereoselective and regioselective synthesis of azepane and azepine derivatives via piperidine ring expansion - Journal of the Chemical Society, Perkin Transactions 1 (RSC Publishing) DOI:10.1039/B204677F
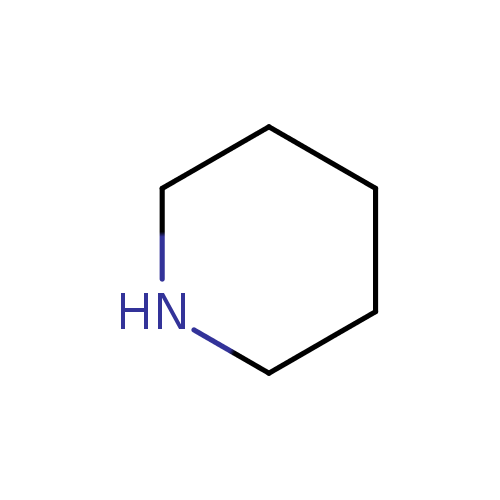
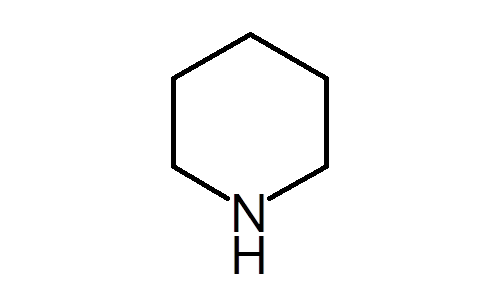
Welcome to Chem Zipper.com......: Pyridine is almost 1 million times less basic than piperidine? Why?
![The Synthesis of Methyl-Substituted Spirocyclic Piperidine-Azetidine (2,7-Diazaspiro[3.5]nonane) and Spirocyclic Piperidine-Pyrrolidine (2,8-Diazaspiro[4.5]decane) Ring Systems | The Journal of Organic Chemistry The Synthesis of Methyl-Substituted Spirocyclic Piperidine-Azetidine (2,7-Diazaspiro[3.5]nonane) and Spirocyclic Piperidine-Pyrrolidine (2,8-Diazaspiro[4.5]decane) Ring Systems | The Journal of Organic Chemistry](https://pubs.acs.org/cms/10.1021/acs.joc.5b02890/asset/images/medium/jo-2015-028905_0009.gif)
The Synthesis of Methyl-Substituted Spirocyclic Piperidine-Azetidine (2,7-Diazaspiro[3.5]nonane) and Spirocyclic Piperidine-Pyrrolidine (2,8-Diazaspiro[4.5]decane) Ring Systems | The Journal of Organic Chemistry